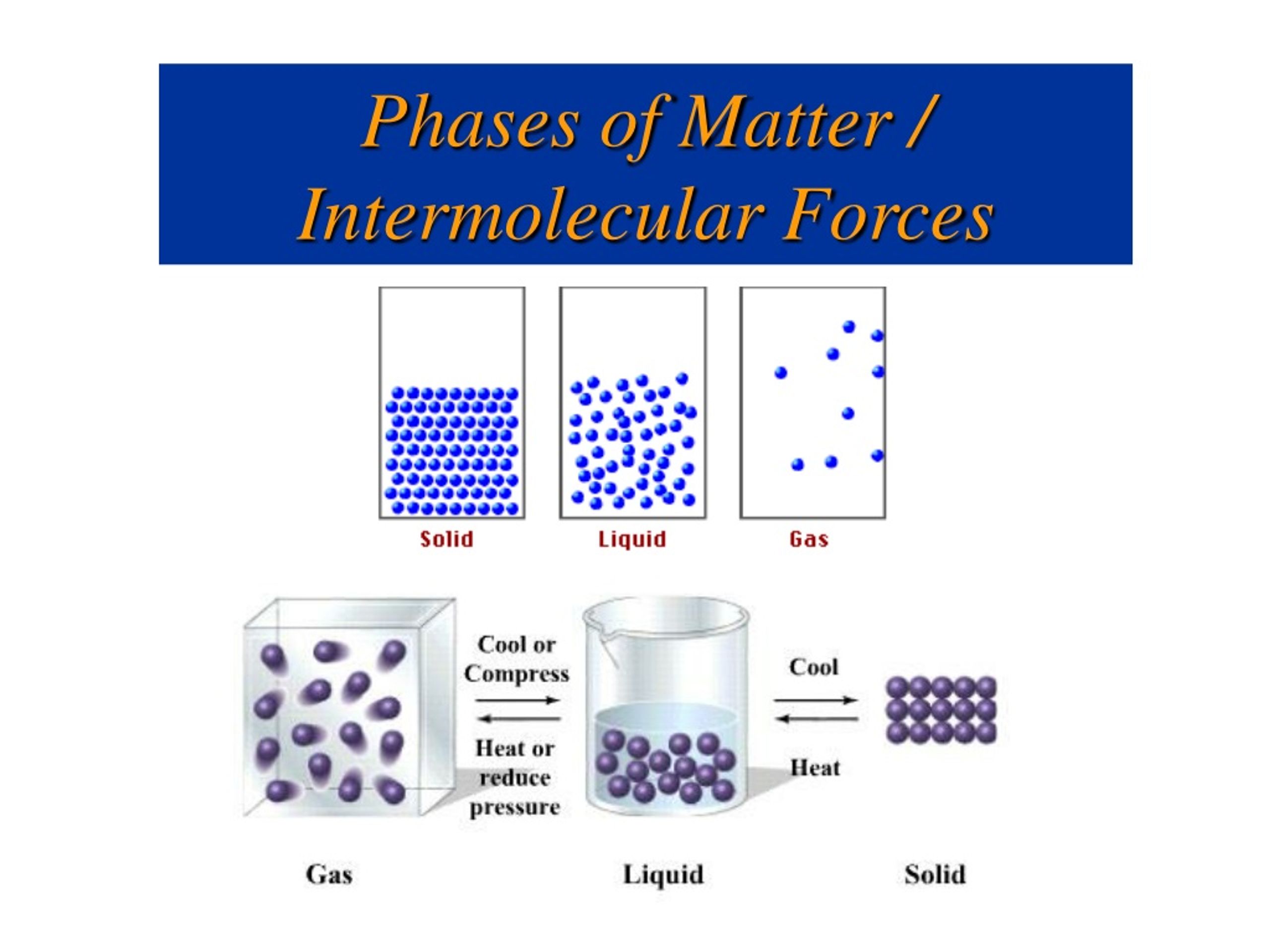
Its particles are still close together but move freely. Matter in the liquid state maintains a fixed volume (assuming no change in temperature or air pressure), but has a variable shape that adapts to fit its container. Matter in the solid state maintains a fixed volume (assuming no change in temperature or air pressure) and shape, with component particles ( atoms, molecules or ions) close together and fixed into place. Historically, the distinction is made based on qualitative differences in properties. 
For a complete list of all exotic states of matter, see the list of states of matter. Many intermediate states are known to exist, such as liquid crystal, and some states only exist under extreme conditions, such as Bose–Einstein condensates and Fermionic condensates (in extreme cold), neutron-degenerate matter (in extreme density), and quark–gluon plasma (at extremely high energy). Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. In physics, a state of matter is one of the distinct forms in which matter can exist.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
